Cardiac action potentials differ from the APs found in other areas of the body.
Typical neural AP duration is around 1ms and those of skeletal muscle are roughly 2-5ms, whereas cardiac action potentials range from 200-400ms.
Nervous and muscle cells (as well as non-pacemaker cardiac cells) use the opening of Na channels to facilitate the depolarisation phase, whereas cardiac pacemaker cells use Ca ions in depolarisation
The transfer of ions from the intracellular environment to the extracellular environment, and vice versa, is what allows for the de and repolarisation of the cardiac muscle cells, and the pacemaker cells. These ions are transferred via channels through the cellular membrane, helping to maintain the differences in charge between the inside and the outside of the cell.
The main forces responsible for facilitating the transfer of ions across the cell membrane are:
- Chemical potential
- causes an ion to move down its concentration gradient
- Electrical potential
- causes an ion to move away from similarly-charged particles
The Transmembrane Potential is the voltage difference between the intra and extracellular environments. A net movement of positive ions out of the cell, causes the TMP to become more negative, and vice versa.
Cardiac ion channels have various properties that enable them to carry out their function:
- Selective
- Only permeable to one specific ion type, based on their structure
- Voltage-sensitive
- Transmembrane potential has to be specific to enable channels to open
- Changing TMP ensures changing opening and closing of channels
- Time-Dependant
- Channels such as Fast Na+, close very soon after opening, and cannot open again until resting potential has been reached.
Non-Pacemaker Action Potentials

- Atrial & ventricular myocytes, Purkinje cells
- Called “fast response cells” due to rapid depolarisation
- Unlike pacemaker cells, they have a true resting potential
Phase 4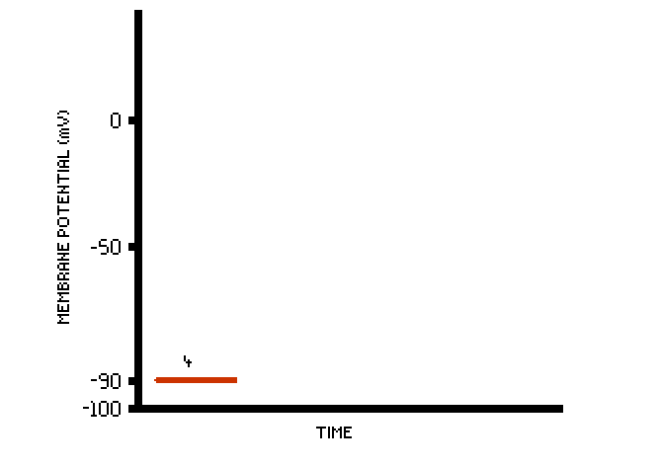
- The quiescent phase
- True resting potential
- -90mV
- K+ channels are open
- Na+ and Ca2 are closed
Phase 0

- Fast depolarization
- AP triggered in neighbouring cell raises the transmembrane potential above its resting -90mV
- Fast Na+ channels open, and Na+ further depolarises the cell
- Once the transmembrane potential reaches -70mV, the cardiomyocyte threshold potential is reached.
- The Na+ current quickly depolarises the transmembrane potential to slightly above 0mv (overshoot), then fast Na+ channels close.
- L-Type Ca2+ open once the transmembrane potential hits -40mV, thereby inducting CA2+ down its concentration gradient
Phase 1

- Being above 0mV, the transmembrane potential is slightly positive
- K+ channels open, expelling K+ and lowering the transmembrane potential to 0mV
Phase 2

- Ca2+ ions are still flowing down the concentration gradient, and into the cell.
- K+ flows down its concentration gradient and out of the cell.
- These two processes are balanced, and maintain the plateau seen at just less than 0mV
Phase 3

- Gradual closing of the Ca2+ channels
- The exiting K+ now outweighs the incoming Ca2+
- This reduces the transmembrane potential back towards the true resting potential of -90mV
Pacemaker Action Potentials

- Found in cells such as the SA node
- Called slow response cells
- Do not utilise Fast Na+ currents
Phase 4

- After Phase 3 of the previous cycle, slow Na+ currents cause the cell membrane to depolarise from -60mV
- At around 50mV, Transient Type Ca++ channels open, and contribute to depolarisation.
- K+ exit from Phase 3 slowing, during this phase
Phase 0

- L-Type Ca++ channels open, depolarising the cell until it reaches its action potential threshold.
- Ca++ channels are slow
Phase 3

- K+ channels open as Ca++ channels close
- Equilibrium potential returns closer to that of K+